The National Agency for Food and Drug Administration and Control (NAFDAC) on Friday alerted the public to the circulation of counterfeit Augmentin 625mg tablets in Nigeria.
According to a statement issued by the agency, the alert followed complaints received by GlaxoSmithKline (GSK), the genuine manufacturer of Augmentin, over suspected fake versions of the product in circulation.
The full statement is published below…
Public Alert No. 024/2026.
Alert on counterfeit Augmentin 625mg Tablets (Batch No. AC3N) in Nigeria
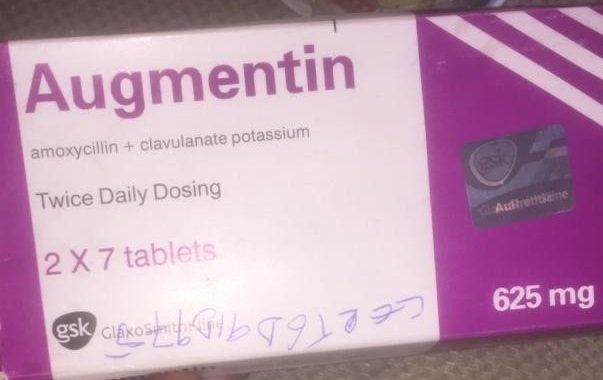
NAFDAC is notifying healthcare providers and the public of the circulation of counterfeit or falsified Augmentin 625mg Tablets (2 x 7 tablets pack) with Batch No. AC3N in Nigeria.
NAFDAC has received information from the Marketing Authorisation Holder for GlaxoSmithKline products in Nigeria regarding suspected counterfeit Augmentin 625mg Tablets.
This alert follows a series of complaints received by the genuine manufacturer, GlaxoSmithKline (GSK), regarding suspected counterfeit Augmentin 625mg Tablets bearing the Batch No. AC3N, circulating in Nigeria.
GSK evaluated and confirmed that the suspected product was counterfeit after reviewing documentary evidence, photographs, and a video from social media. Although the product shared the same batch number as authentic stock previously supplied to Nigeria, it exhibited inconsistent manufacturing and expiry dates, indicating that it had been falsified.
Findings from the manufacturer’s investigation:
• Batch AC3N was manufactured at Worthing, United Kingdom, on 23 August 2023.
• The batch was packed between 19 and 20 September 2023.
• No batch with the number AC3N was produced in September 2025.
• The manufacturing and expiry dates shown on the suspect sample (Manufactured: September 2025 / Expiry: September 2028) are inconsistent with GSK production records and cannot be attributed to a GSK-produced batch.
• Packaging-level discrepancies, including textual errors and poor fin seal quality, were identified.
• Based on the evidence reviewed, the product was assessed as counterfeit.
Augmentin (amoxicillin/clavulanic acid) is an antibiotic used for the treatment of bacterial infections. Any falsified version may contain incorrect amounts of active ingredients, no active ingredient, or harmful substances.
Product Details:
• Product Name: Augmentin 625mg Tablets
• Pack Size: 2 x 7 Tablets
• Batch Number: AC3N
• Manufacturer of Genuine Product: GlaxoSmithKline (GSK), Worthing Site, United Kingdom.
Healthcare professionals are advised to review stock in hospitals, clinics, and pharmacy stores, immediately quarantine any suspected counterfeit stock, and discontinue the sale or distribution of any suspected counterfeit.
Report any suspected falsified or compromised products (medicines or medical devices) to the nearest NAFDAC office, call 0800-162-3322, or send an email to sf.alert@nafdac.gov.ng




Leave a Reply