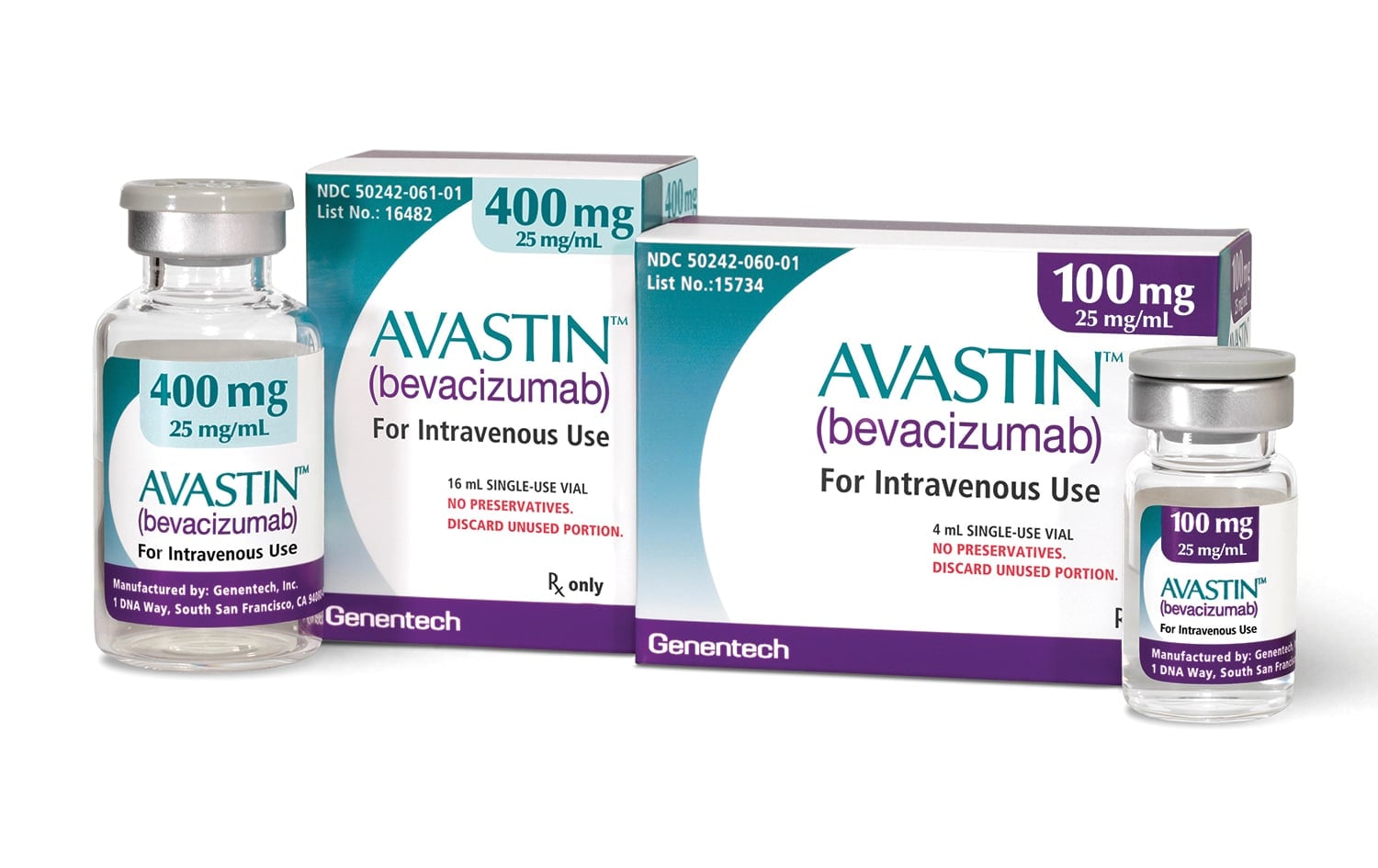
The National Agency for Food and Drug Administration and Control (NAFDAC) has warned Nigerians about the circulation of counterfeit cancer medicines, Avastin and Tecentriq, in the country.
Avastin (bevacizumab) is used for the treatment of recurrent glioblastoma in adults.
Tecentriq (atezolizumab) is a prescription immunotherapy used to treat several advanced cancers, including non-small cell lung cancer (NSCLC), extensive-stage small cell lung cancer (ES-SCLC), hepatocellular carcinoma (liver cancer), and melanoma.
In a public alert, the agency said it is “notifying healthcare professionals and the public of confirmed counterfeit batches of Avastin 400mg/16ml (Bevacizumab) and Tecentriq 1200mg/20ml (Atezolizumab) circulating in Nigeria”.
The alert, according to NAFDAC, followed official communication from Roche Nigeria, which confirmed several cases of counterfeit products in Nigeria after receiving complaints from healthcare professionals across the country.
The agency said in many cases, patients brought suspected counterfeit products to health facilities, adding that the medicines were sold at significantly lower prices, ranging from N180,000 to N350,000.
“The Marketing Authorization Holder (MAH) reported receiving complaints from healthcare professionals across Nigeria,” the alert reads.
“On investigation, the MAH (Roche Nigeria) compared the complaint samples to the genuine retained samples. The findings have provided clear evidence of counterfeit packaging material of the reported products.”
The agency listed discrepancies observed during the investigation, noting that “batch numbers do not correspond to genuine Roche batches”, alongside “differences in artwork and printing quality”, “wrong placement of text”, and “incorrect variable data”.
It added that “tamper-evident labels” and serial numbers on the products were also inconsistent with Roche’s standard system.
NAFDAC identified specific counterfeit batches of Avastin 400mg/16ml with batch numbers H4239A70, H2290A34, and A3508B02, as well as Tecentriq 1200mg/20ml with batch number B3071A12.
The agency, however, said chemical analysis of the products could not be carried out as “the investigation depended on packaging photographs sent by the complainants; no physical samples were available”.
NAFDAC warned that counterfeit oncology medicines “may contain incorrect or no active ingredients, harmful contaminants, or incorrect dosage strength”, which could lead to “treatment failure, disease progression, serious adverse events, or or death”.
It said all zonal directors and state coordinators had been directed to intensify surveillance and “mop up the counterfeit product within the zones and states”.
NAFDAC also urged importers, distributors, healthcare professionals and patients to remain vigilant and ensure that medicines are sourced only from authorised suppliers.
The agency advised the public to report suspected cases of substandard or falsified medicines through its offices, hotline, or official reporting platforms.



Leave a Reply