Chidi Samuel| Nigeria on Friday confirmed 212 new cases of #COVID19 bringing the country’s virus load to 61, 194.
The Nigeria Centre For Disease Control which made the disclosure in a statement via its verified Twitter handle said the new cases were recorded in eight states and the FCT.
According the agency’s data, Lagos recorded -85 new infections, followed by Oyo-72.
Other states with new infections are, FCT-21
Ogun-11, Plateau-11, Katsina-6, Kaduna-5
and Osun-1.
The NCDC further noted that so far, the country has recorded 52,304 recoveries with 1,119 deaths.
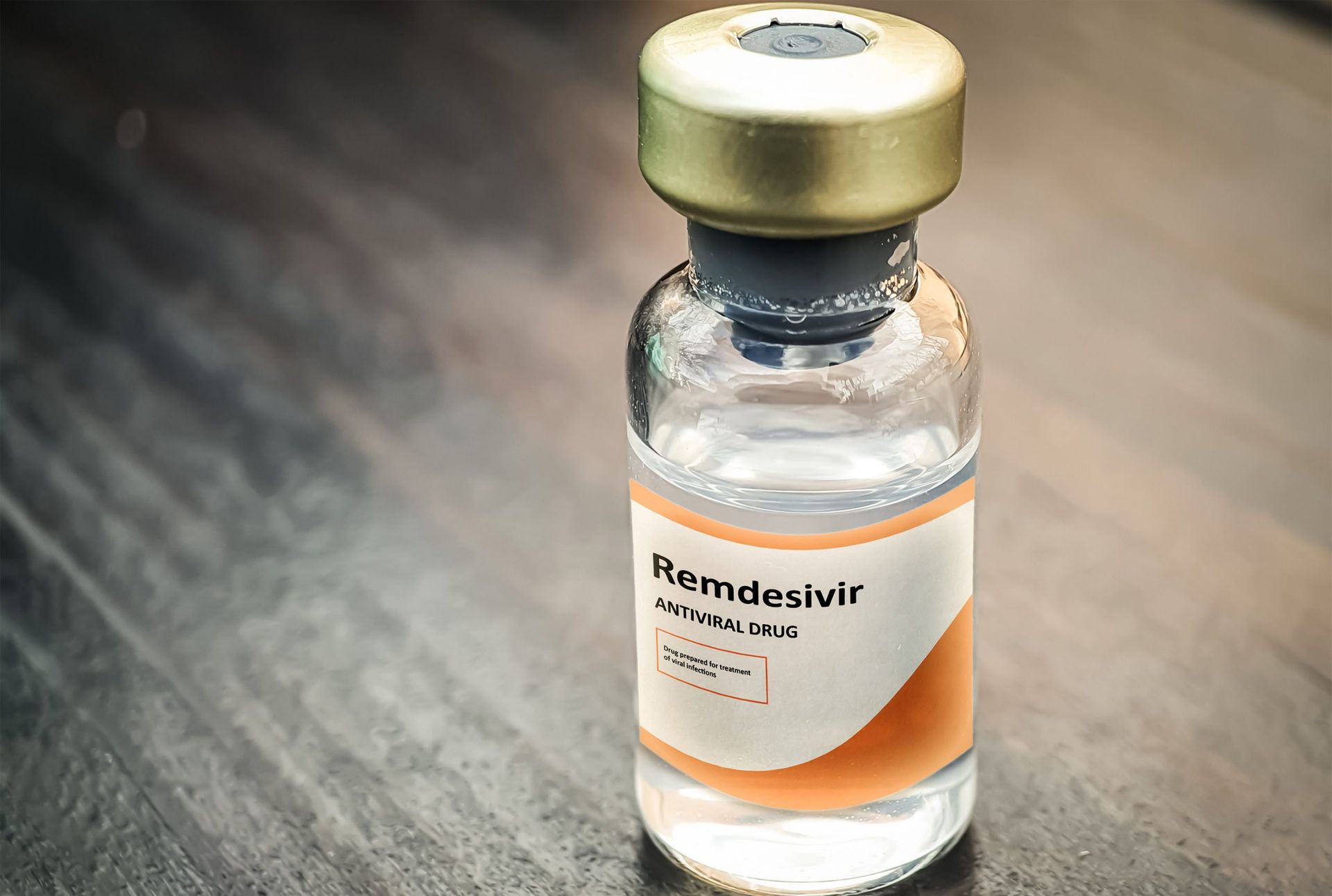
‘Remdesivir has little effect on COVID-19’ — WHO
Meanwhile, the World Health Organisation (WHO) says remdesivir, an antiviral drug considered as one of the treatments for COVID-19, has “little or no effect” on mortality or reduction in hospital stays for patients.
This is according to the data from WHO’s solidarity trial.
The trial, conducted among 11,000 patients in 30 countries, involved four potential treatments — remdesivir, hydroxychloroquine, lopinavir/ritonavir, HIV drug combination and interferon.
WHO, in a statement on Thursday, said the results of the trial which are yet to be reviewed suggest that remdesivir and three other existing medications for the treatment of COVID-19 have no substantial effect on mortality or on the length of time spent in the hospital.
Despite questions about its effectiveness, remdesivir was the first drug to receive the approval of the European Commission in the treatment of severe cases of COVID-19 in July.
The EU had signed a contract with Gilead Sciences, US biopharmaceutical company, for 30,000 doses of the drug on behalf of EU member states.
In April, Gilead had announced that the drug was found to be effective on COVID-19 patients during its clinical trial which began in February, adding that it administered the drug on 397 severely ill patients and more than half of them were discharged within two weeks.
But WHO’s result contradicts a recent study conducted by Gilead, which concluded that patients receiving remdesivir recovered five days faster, and in patients with severe disease, seven days faster than those given a placebo.
The trial involved 1,060 hospitalised patients worldwide.
The pharmaceutical company also said the drug reduces the likelihood of patients progressing to severe stages of the disease and that it showed significant reduction in mortality.
However, WHO noted that newer antiviral drugs, immunomodulators and anti-SARS COV-2 monoclonal antibodies are now being considered for evaluation for new COVID-19 treatment options.
With close to 40 million confirmed COVID-19 cases globally, scientists are working to develop vaccines and treatments to fight the pandemic and eventually flatten the curve of infection.
With agency report